Princess Leia brainwaves may help you learn in your sleep
Watch a baby sea turtle being hypnotised so we can weigh it
Could a vast rubber boom clean up tonnes of ocean plastic?
Tourists pick up antibiotic-resistance genes in just two days
Watch NASA Set a Fire in Space … on Purpose

Who needs water? Dry volcanic vents more alive than wet soil
Scientists Are Trying to Make Nuclear Fusion With Frickin’ Lasers

Are you ready for mood-altering drugs precisely inserted into your brain?
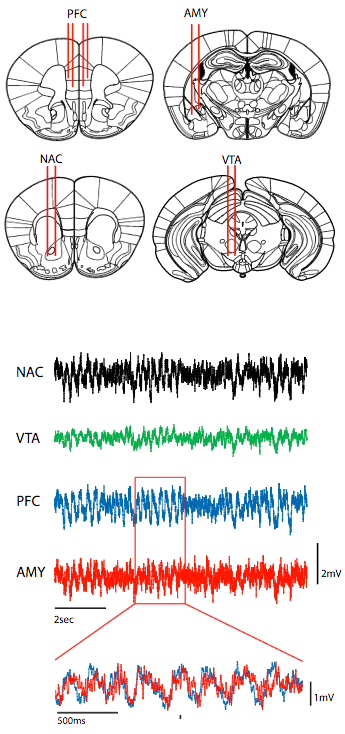
To directly probe the relationship between prefrontal cortex circuitry (PFC), limbic network oscillatory dysfunction, and the emergence of depression-related behavior, the researchers implanted mice with microwire recording electrodes in PFC and three relevant limbic brain regions implicated in major depressive disorder: nucleus accumbens (NAC), amygdala (AMY), and ventral tegmental area (VTA). Shown below: representative local field potential traces of neural signals. In the overlaid traces below, note that PFC oscillations (blue) tended to precede AMY oscillations (red). (credit: Rainbo Hultman et al./Neuron)
Imagine if doctors could precisely insert a tiny amount of a custom drug into a specific circuit in your brain and improve your depression (or other mood problems) — instead of treating the entire brain.
That’s exactly what Duke University researchers have explored in mice. Stress-susceptible animals that appeared depressed or anxious were restored to relatively normal behavior this way, according to a study appearing in the forthcoming July 20 issue of Neuron.
The plan was to define specific glitches in the neural circuits and then use a drug to fix them. The ambitious goal: go from a protein, to a signaling activity, to a cell, to a circuit, to activity that happens across the whole brain, to actual behavior.
1. Identify the key neurons in the prefrontal cortex
The researchers first determined how the prefrontal cortex is used as a pacemaker for the limbic system, said lead researcher Kafui Dzirasa, an assistant professor of psychiatry and behavioral sciences, and neurobiology.
The team started by precisely placing arrays of 32 electrodes in four brain areas of the mice (see illustration above). Then they recorded brain activity as these mice were subjected to a stressful situation called chronic social defeat.* This allowed the researchers to observe the activity between the prefrontal cortex and three areas of the limbic system that are implicated in major depression.
To interpret the complicated data coming from the electrodes, the team used machine learning algorithms — identifying which parts of the data seemed to be the timing control signal between the prefrontal cortex and the limbic system— and then zeroed in on the individual neurons involved in that cortical signal and its corresponding circuit.
2. Inject a drug to restore function
They then applied engineered molecules called DREADD (Designer Receptors Exclusively Activated by Designer Drug), developed by University of North Carolina at Chapel Hill pharmacologist Bryan Roth, in very tiny amounts (0.5 microliter). A drug that attaches only to that DREADD is then administered to give the researchers control over the circuit.
They found that direct stimulation of PFC-amygdala neural circuitry with DREADDs normalized PFC-dependent limbic synchrony in stress-susceptible animals and restored normal behavior.
The researchers suggest that their findings also demonstrate an interdisciplinary approach that can be used to identify the large-scale network changes that underlie complex emotional pathologies and the specific network nodes that can be used to develop targeted interventions.
“These subcortical circuits are the key regulators of our emotional life,” said Helen Mayberg, a professor of psychiatry, neurology and radiology at Emory University who was not involved in this research. “What’s great about this paper is that they use different approaches to see a circuit that’s relevant to a lot of disorders,” said Mayberg, who has been pioneering deep-brain stimulation of very specific sites in the human prefrontal cortex to treat mood disorders.
But she cautions that to assess anything like “mood” in a mouse, one can only infer from its behaviors. “It’s hard to do, even in a human,” she said.
This work was supported by funding from National Institutes of Mental Health and a research incubator award from the Duke Institute for Brain Sciences.
* The mice were repeatedly exposed to larger aggressive animals for 10–15 consecutive days. At the end of this protocol, animals exhibit multiple depressive endophenotypes including hedonic dysfunction, circadian dysregulation, anxiety, and psychomotor retardation.
Abstract of Dysregulation of Prefrontal Cortex-Mediated Slow-Evolving Limbic Dynamics Drives Stress-Induced Emotional Pathology
Circuits distributed across cortico-limbic brain regions compose the networks that mediate emotional behavior. The prefrontal cortex (PFC) regulates ultraslow (<1 Hz) dynamics across these networks, and PFC dysfunction is implicated in stress-related illnesses including major depressive disorder (MDD). To uncover the mechanism whereby stress-induced changes in PFC circuitry alter emotional networks to yield pathology, we used a multi-disciplinary approach including in vivo recordings in mice and chronic social defeat stress. Our network model, inferred using machine learning, linked stress-induced behavioral pathology to the capacity of PFC to synchronize amygdala and VTA activity. Direct stimulation of PFC-amygdala circuitry with DREADDs normalized PFC-dependent limbic synchrony in stress-susceptible animals and restored normal behavior. In addition to providing insights into MDD mechanisms, our findings demonstrate an interdisciplinary approach that can be used to identify the large-scale network changes that underlie complex emotional pathologies and the specific network nodes that can be used to develop targeted interventions.
How exercise improves memory
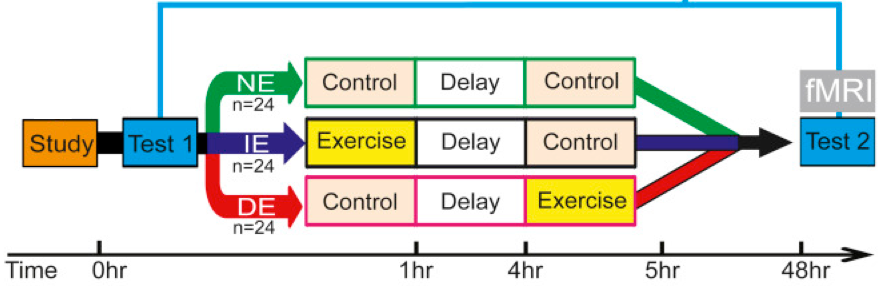
Experimental design (partial view). Study participants learned 90 picture-location associations over a period of approximately 40 minutes before being randomly assigned to one of three groups: one group performed exercise immediately, the second performed exercise four hours later, and the third did not perform any exercise. Forty-eight hours later, participants returned for a test to show how much they remembered while their brains were imaged via magnetic resonance imaging (MRI).
Physical exercise after learning improves memory and memory traces if the exercise is done four hours later, and not immediately after learning, according to findings recently reported (open-access) in the Cell Press journal Current Biology.
It’s not yet clear exactly how or why delayed exercise has this effect on memory. However, earlier studies of laboratory animals suggest that naturally occurring chemical compounds in the body known as catecholamines, including dopamine and norepinephrine, can improve memory consolidation, say the researchers at the Donders Institute at the Radboud University Medical Center in the Netherlands. One way to boost catecholamines is through physical exercise.
The researchers tested the effects of a single session of physical exercise after learning on memory consolidation and long-term memory. Seventy-two study participants learned 90 picture-location associations over a period of approximately 40 minutes before being randomly assigned to one of three groups: one group performed exercise immediately, the second performed exercise four hours later, and the third did not perform any exercise. The exercise consisted of 35 minutes of interval training on an exercise bike at an intensity of up to 80 percent of participants’ maximum heart rates. Forty-eight hours later, participants returned for a test to show how much they remembered while their brains were imaged via magnetic resonance imaging (MRI). The researchers found that those who exercised four hours after their learning session retained the information better two days later than those who exercised either immediately or not at all.
The researchers plan to follow up with another study of the timing and molecular underpinnings of exercise and its influence on learning and memory in more detail.
The researchers were supported by a grant from the European Research Council.
Brain chemicals

Exercise induces synthesis of a chemical called DBHB in the liver. In the hippocampus, DBHB induces Bdnf gene expression, which in turn has positive effects on memory, cognition and synaptic transmission. (credit: Sama F. Sleiman et al./eLife)
In a related study published in eLife June 2 with mice, researchers note that exercise is known to be accompanied by an increase in brain-derived neurotrophic factor (BDNF) in the hippocampus, which is associated with cognitive improvement and the alleviation of depression and anxiety.
But how? It is known that a substance known as β-hydroxybutyrate (DBHB), produced in the liver from fatty acids, serves as an alternative energy source when glucose (blood sugar) levels are reduced. In their research (with mice on a running wheel for 30 days vs. no exercise), they found that the resulting increase of DBHB blocked the action of histone enzymes, which normally inhibit the production of BDNF.
Confirming that, injecting DBHB directly into the brains of mice also led to increase in BDNF.
Abstract of Physical Exercise Performed Four Hours after Learning Improves Memory Retention and Increases Hippocampal Pattern Similarity during Retrieval
Persistent long-term memory depends on successful stabilization and integration of new memories after initial encoding [ 1, 2 ]. This consolidation process is thought to require neuromodulatory factors such as dopamine, noradrenaline, and brain-derived neurotrophic factor [ 3–7 ]. Without the release of such factors around the time of encoding, memories will decay rapidly [ 3, 5, 6, 8 ]. Recent studies have shown that physical exercise acutely stimulates the release of several consolidation-promoting factors in humans [ 9–14 ], raising the question of whether physical exercise can be used to improve memory retention [ 15–17 ]. Here, we used a single session of physical exercise after learning to exogenously boost memory consolidation and thus long-term memory. Three groups of randomly assigned participants first encoded a set of picture-location associations. Afterward, one group performed exercise immediately, one 4 hr later, and the third did not perform any exercise. Participants otherwise underwent exactly the same procedures to control for potential experimental confounds. Forty-eight hours later, participants returned for a cued-recall test in a magnetic resonance scanner. With this design, we could investigate the impact of acute exercise on memory consolidation and retrieval-related neural processing. We found that performing exercise 4 hr, but not immediately, after encoding improved the retention of picture-location associations compared to the no-exercise control group. Moreover, performing exercise after a delay was associated with increased hippocampal pattern similarity for correct responses during delayed retrieval. Our results suggest that appropriately timed physical exercise can improve long-term memory and highlight the potential of exercise as an intervention in educational and clinical settings.
Abstract of Exercise promotes the expression of brain derived neurotrophic factor (BDNF) through the action of the ketone body β-hydroxybutyrate
Exercise induces beneficial responses in the brain, which is accompanied by an increase in BDNF, a trophic factor associated with cognitive improvement and the alleviation of depression and anxiety. However, the exact mechanisms whereby physical exercise produces an induction in brain Bdnf gene expression are not well understood. While pharmacological doses of HDAC inhibitors exert positive effects on Bdnf gene transcription, the inhibitors represent small molecules that do not occur in vivo. Here, we report that an endogenous molecule released after exercise is capable of inducing key promoters of the Mus musculus Bdnf gene. The metabolite β-hydroxybutyrate, which increases after prolonged exercise, induces the activities of Bdnf promoters, particularly promoter I, which is activity-dependent. We have discovered that the action of β-hydroxybutyrate is specifically upon HDAC2 and HDAC3, which act upon selective Bdnf promoters. Moreover, the effects upon hippocampal Bdnfexpression were observed after direct ventricular application of β-hydroxybutyrate. Electrophysiological measurements indicate that β-hydroxybutyrate causes an increase in neurotransmitter release, which is dependent upon the TrkB receptor. These results reveal an endogenous mechanism to explain how physical exercise leads to the induction of BDNF.
