
New software from Mapbox could make it easier to turn geodata into useful apps and maps.
The post This Software Could Change How the Maps in Your Apps Get Made appeared first on WIRED.

Science and reality

New software from Mapbox could make it easier to turn geodata into useful apps and maps.
The post This Software Could Change How the Maps in Your Apps Get Made appeared first on WIRED.
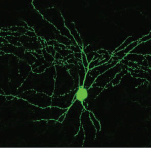
PV+ interneuron (credit: Nathalie Dehorter et al./Science)
King’s College London researchers have developed a new molecular “switch” that controls the properties of certain neurons in response to changes in the activity of their neural network — suggesting that these circuits in our brain are tuneable and could have implications that go far beyond basic neuroscience.
The researchers, from the MRC Centre for Developmental Neurobiology (MRC CDN) at the Institute of Psychiatry, Psychology & Neuroscience (IoPPN), led by Professor Oscar Marín, have discovered that some neurons in the cerebral cortex can adapt their properties in response to changes in network activity, such as learning a motor (muscle) task.
The authors studied two apparently different classes of fast-spiking interneurons but discovered they were actually looking at the same neuron — one with the ability to oscillate between two different ground states. The authors then identified the molecular factor responsible for tuning the properties of these cells: a transcription factor (a protein able to influence gene expression) known as Er81.
Neurons with “tremendous plasticity”
Fast-spiking interneurons, known as FS PV+, are members of a general class of neurons whose primary role is regulating the activity of pyramidal cells (the principal cells of the cerebral cortex). These PV+ interneurons play a prominent role in the regulation of plasticity and learning.
The researchers believe that PV+ interneurons take on properties based on how they adapt and respond to internal and external influences to encode information. “In other words, that our [brain's] ‘hardware’ is tuneable, at least to some extent,” said Nathalie Dehorter of the MRC CDN and first author of the study, published in the journal Science on Sept. 11, 2015.
“Our study demonstrates the tremendous plasticity of the brain, and how this relates to fundamental processes such as learning,” said Marín. “Understanding the mechanisms that regulate this plasticity, and why it tends to dissipate when we age, has enormous implications that go beyond fundamental neuroscience, from informing education policies to developing new therapies for neurological disorders such as epilepsy.”
Abstract of Tuning of fast-spiking interneuron properties by an activity-dependent transcriptional switch
The function of neural circuits depends on the generation of specific classes of neurons. Neural identity is typically established near the time when neurons exit the cell cycle to become postmitotic cells, and it is generally accepted that, once the identity of a neuron has been established, its fate is maintained throughout life. Here, we show that network activity dynamically modulates the properties of fast-spiking (FS) interneurons through the postmitotic expression of the transcriptional regulator Er81. In the adult cortex, Er81 protein levels define a spectrum of FS basket cells with different properties, whose relative proportions are, however, continuously adjusted in response to neuronal activity. Our findings therefore suggest that interneuron properties are malleable in the adult cortex, at least to a certain extent.

A tide gauge less than 100 miles from the quake's epicenter has logged wave heights in excess of 14 feet.
The post The Chile Earthquake’s First Tsunami Waves Strike appeared first on WIRED.

Artist’s representation of a calcium ion channel affected by OptoSTIM1 (credit: Institute for Basic Science)
An advanced process for precision control of cellular calcium ion (Ca2+) channels in living organisms has been engineered by a research team at the Korea Advanced Institute of Science and Technology (KAIST) and the IBS Center for Cognition and Sociality.
Calcium ions are a crucial part of diverse cellular functions such as contraction, excitation, growth, differentiation and death. Severe Ca2+ deficiency is linked to cardiac arrhythmia, cognitive impairment, and ataxia.
The new process uses optogenetics, or control of cells by light. The researchers added a new light-sensitive, plant-human hybrid protein to cells to efficiently modulate calcium ion channels in cells by shining blue light on them.
The hybrid protein combines a photoreceptor protein called cryptochrome 2 (Cry2) from a small, flowering plant Arabidopsis thaliana with the STromal Interaction Molecule 1 (STIM1), a protein found in almost all animals that opens cellular Ca2+ channels.
They named the resultant hybrid molecule OptoSTIM1.
When they shined blue light on the OptoSTIM1-expressing cells, they were able to coax them to open their Ca2+ channels and allow an influx of 5 to 10 times more Ca2+ than in previous studies.
Increasing learning capacity in mice
To test the functional effect of the Ca2+ influx, the IBS team introduced OptoSTIM1 to the hippocampus of a living mouse. They compared sets of light-illuminated mice to non-illuminated mice expressing OptoSTIM1 in an environment in which they introduced a conditioning cue followed by a fear stimulus.
In subsequent tests they observed that light-illuminated mice with the OptoSTIM1 expression had a nearly twofold increase in fear response when placed in the testing environment without the conditioning cue than the non-light-stimulated mice. That indicated that the OptoSTIM1 expression (and resultant Ca2+ uptake) was an effective method for memory enhancement.
Neurological enhancements and treatments
The researchers say this work opens the door for future research into optogenetically enhanced memory and learning studies and into treating neurological diseases that are a result of a dysfunction in Ca2+ regulation.
This may also be a step towards discovering applications for drugs as well as therapeutic Ca2+ modulation. According to Kyung, “There are diseases that result from dysfunction in cellular Ca2+ regulation, such as Alzheimer’s disease, so we can apply our system to those areas and hopefully in the near future help people to recover from those diseases.”
This may also allow for future non-invasive and non-drug treatments or may help to mitigate and eventually cure some neurological diseases.
Team is led by Won Do Heo, associate professor together with Professor Yong-Mahn Han and Professor Daesoo Kim.
Abstract of Optogenetic control of endogenous Ca2+ channels in vivo
Calcium (Ca2+) signals that are precisely modulated in space and time mediate a myriad of cellular processes, including contraction, excitation, growth, differentiation and apoptosis. However, study of Ca2+ responses has been hampered by technological limitations of existing Ca2+-modulating tools. Here we present OptoSTIM1, an optogenetic tool for manipulating intracellular Ca2+ levels through activation of Ca2+-selective endogenous Ca2+ release−activated Ca2+ (CRAC) channels. Using OptoSTIM1, which combines a plant photoreceptor and the CRAC channel regulator STIM1 (ref. 4), we quantitatively and qualitatively controlled intracellular Ca2+ levels in various biological systems, including zebrafish embryos and human embryonic stem cells. We demonstrate that activating OptoSTIM1 in the CA1 hippocampal region of mice selectively reinforced contextual memory formation. The broad utility of OptoSTIM1 will expand our mechanistic understanding of numerous Ca2+-associated processes and facilitate screening for drug candidates that antagonize Ca2+ signals.

An 8.3 moment magnitude quake struck northwest of Santiago.
The post A Massive Quake Just Struck Off the Coast of Chile appeared first on WIRED.

The oil and gas giant took climate change seriously.
The post Exxon’s Own Scientists Confirmed Climate Change—In the 70s appeared first on WIRED.

The worm turns: injecting a transgenic nematode worm with tyramine induces the worm to switch from forward locomotion (dashed red line, starting at X) to backward locomotion (dashed blue line) (credit: Jennifer K. Pirri et al./PLOS Biology)
In 1963, Yale professor of physiology and psychiatry Dr. Jose Delgado implanted an stimulating electrode in the caudate nucleus of a fighting bull, bravely jumped into the bullring, and stopped the animal in its tracks by remotely activating the electrode. Now UMass Medical School scientists have taken neural control precision down to the synapse level, reversing a C. elegans (nematode) worm’s head position or locomotion direction by simply switching one of its synapses (neuron-to-neuron connections) from inhibitory to excitatory (“off” to “on”).
The research, published in an open-access paper in PLOS Biology, offers a new approach for studying the neural circuits that govern behavior and has implications for refining the connectome (neural roadmap), which is important for understanding how the 100 billion neurons and quadrillion (1015) synapse process information and control behavior.
Currently, the connectome doesn’t include information about the activity of specific neurons or the signals they transmit. The complexity of the human brain makes it almost impossible to address questions such as how stable are neural circuits in the brain and how their wiring constrains the flow of information or the behaviors they control.
So Mark Alkema, PhD, associate professor of neurobiology, turned to the nematode C. elegans. A tiny worm with only 302 neurons, it is the only animal whose neural roadmap has been completely defined.
Worm mind control
Alkema and colleagues wanted to determine if flipping the sign (inhibitory or excitatory) of a synapse in the worm’s brain was enough to reverse a behavior. To do this, they analyzed the touch response that allows C. elegans to escape from carnivorous fungi that use threadlike nooses to catch nematodes. During this escape response, neurotransmitters in C. elegans are released that activate an inhibitory ion channel. This causes the worm to relax its head and quickly reverse direction away from the predator.
Synapse firing is determined by the charge of the ions that flow through channels. So they replaced the inhibitory ion channel with an excitatory version of the channel in a live nematode. “Surprisingly, the engineered channel does not affect the worms’ development and is properly incorporated into the neural circuits of the worm brain,” said Alkema. “Cells that are normally inhibited in the brain now get activated.”
“What was most striking is that we were able to completely reverse behavior by simply switching the sign of a synapse in the neural network,” explained Alkema. “Now the animal contracts its head and tends to move forward in response to touch. This suggests that the neural wiring diagram is remarkably stable and allows these types of changes.”
“Our studies indicate that switching the sign of a synapse not only provides a novel synthetic mechanism to flip behavioral output but could even be an evolutionary mechanism to change behavior,” said Alkema. “As we start to unravel the complexity and design of the neural network, it holds great promise as a novel mechanism to test circuit function or even design new neural circuits in vivo.”
Abstract of A Change in the Ion Selectivity of Ligand-Gated Ion Channels Provides a Mechanism to Switch Behavior
Behavioral output of neural networks depends on a delicate balance between excitatory and inhibitory synaptic connections. However, it is not known whether network formation and stability is constrained by the sign of synaptic connections between neurons within the network. Here we show that switching the sign of a synapse within a neural circuit can reverse the behavioral output. The inhibitory tyramine-gated chloride channel, LGC-55, induces head relaxation and inhibits forward locomotion during the Caenorhabditis elegans escape response. We switched the ion selectivity of an inhibitory LGC-55 anion channel to an excitatory LGC-55 cation channel. The engineered cation channel is properly trafficked in the native neural circuit and results in behavioral responses that are opposite to those produced by activation of the LGC-55 anion channel. Our findings indicate that switches in ion selectivity of ligand-gated ion channels (LGICs) do not affect network connectivity or stability and may provide an evolutionary and a synthetic mechanism to change behavior.

When a volcano becomes restless, the language we use to describe that unrest is different. Can we all speak the same language?
The post How Can You Tell if a Volcano Is Restless? It’s Complicated appeared first on WIRED.