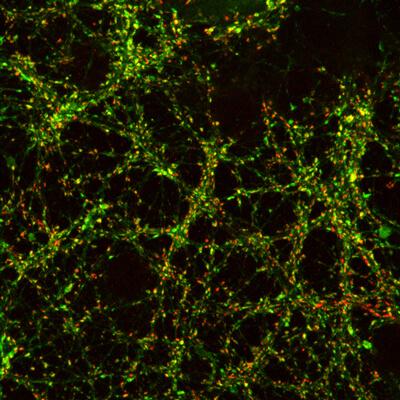
Neurons created from chemically induced neural stem cells. The cells were created from skin cells that were reprogrammed into neural stem cells using a cocktail of only nine chemicals. This is the first time cellular reprogramming has been accomplished without adding external genes to the cells. (credit: Mingliang Zhang, PhD, Gladstone Institutes)
Scientists at the Gladstone Institutes have used chemicals to transform skin cells into heart cells and brain cells, instead of adding external genes — making this accomplishment a breakthrough, according to the scientists.
The research lays the groundwork for one day being able to regenerate lost or damaged cells directly with pharmaceutical drugs — a more efficient and reliable method to reprogram cells and one that avoids medical concerns surrounding genetic engineering.
Instead, in two studies published in an open-access paper in Science and in Cell Stem Cell, the team of scientists at the Roddenberry Center for Stem Cell Biology and Medicine at Gladstone used chemical cocktails to gradually coax skin cells to change into organ-specific stem-cell-like cells and ultimately into heart or brain cells.
“This method brings us closer to being able to generate new cells at the site of injury in patients,” said Gladstone senior investigator Sheng Ding, PhD, the senior author on both studies. “Our hope is to one day treat diseases like heart failure or Parkinson’s disease with drugs that help the heart and brain regenerate damaged areas from their own existing tissue cells. This process is much closer to the natural regeneration that happens in animals like newts and salamanders, which has long fascinated us.”
Chemically Repaired Hearts
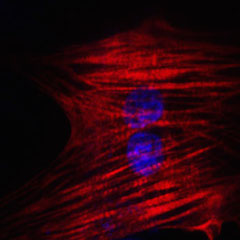
A human heart cell that was chemically reprogrammed from a human skin cell (credit: Nan Cao/Gladstone Institutes)
Transplanted adult heart cells do not survive or integrate properly into the heart and few stem cells can be coaxed into becoming heart cells.
Instead, in the Science study, the researchers used a cocktail of nine chemicals to change human skin cells into beating heart cells. By trial and error, they found the best combination of chemicals to begin the process by changing the cells into a state resembling multipotent stem cells (cells that can turn into many different types of cells in a particular organ). A second cocktail of chemicals and growth factors then helped transition the cells to become heart muscle cells.
With this method, more than 97% of the cells began beating, a characteristic of fully developed, healthy heart cells. The cells also responded appropriately to hormones, and molecularly, they resembled heart muscle cells, not skin cells. What’s more, when the cells were transplanted into a mouse heart early in the process, they developed into healthy-looking heart muscle cells within the organ.
“The ultimate goal in treating heart failure is a robust, reliable way for the heart to create new muscle cells,” said Srivastava, co-senior author on the Science paper. “Reprogramming a patient’s own cells could provide the safest and most efficient way to regenerate dying or diseased heart muscle.”
Rejuvenating the brain with neural stem cell-like cells
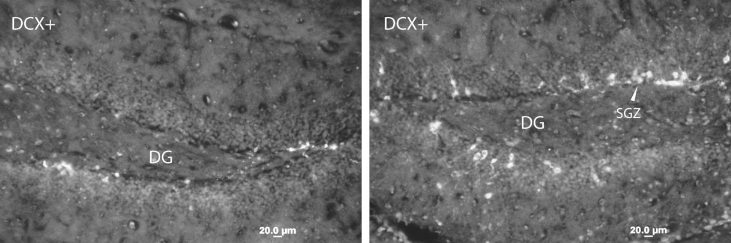
In the second study, authored by Gladstone postdoctoral scholar Mingliang Zhang, PhD, and published in Cell Stem Cell, the scientists created neural stem-cell-like cells from mouse skin cells using a similar approach.
The chemical cocktail again consisted of nine molecules, some of which overlapped with those used in the first study. Over ten days, the cocktail changed the identity of the cells, until all of the skin-cell genes were turned off and the genes of the neural stem-cell-like cells were gradually turned on.
When transplanted into mice, the neural stem-cell-like cells spontaneously developed into the three basic types of brain cells: neurons, oligodendrocytes, and astrocytes. The neural stem-cell-like cells were also able to self-replicate, making them ideal for treating neurodegenerative diseases or brain injury.
With their improved safety, these neural stem-cell-like cells could one day be used for cell replacement therapy in neurodegenerative diseases like Parkinson’s disease and Alzheimer’s disease, according to co-senior author Yadong Huang, MD, PhD, a senior investigator at Gladstone. “In the future, we could even imagine treating patients with a drug cocktail that acts on the brain or spinal cord, rejuvenating cells in the brain in real time.”
Gladstone Institutes | Chemically Reprogrammed Beating Heart Cell
Abstract of Conversion of human fibroblasts into functional cardiomyocytes by small molecules
Reprogramming somatic fibroblasts into alternative lineages would provide a promising source of cells for regenerative therapy. However, transdifferentiating human cells to specific homogeneous, functional cell types is challenging. Here we show that cardiomyocyte-like cells can be generated by treating human fibroblasts with a combination of nine compounds (9C). The chemically induced cardiomyocyte-like cells (ciCMs) uniformly contracted and resembled human cardiomyocytes in their transcriptome, epigenetic, and electrophysiological properties. 9C treatment of human fibroblasts resulted in a more open-chromatin conformation at key heart developmental genes, enabling their promoters/enhancers to bind effectors of major cardiogenic signals. When transplanted into infarcted mouse hearts, 9C-treated fibroblasts were efficiently converted to ciCMs. This pharmacological approach for lineage-specific reprogramming may have many important therapeutic implications after further optimization to generate mature cardiac cells.
Abstract of Pharmacological Reprogramming of Fibroblasts into Neural Stem Cells by Signaling-Directed Transcriptional Activation
Cellular reprogramming using chemically defined conditions, without genetic manipulation, is a promising approach for generating clinically relevant cell types for regenerative medicine and drug discovery. However, small-molecule approaches for inducing lineage-specific stem cells from somatic cells across lineage boundaries have been challenging. Here, we report highly efficient reprogramming of mouse fibroblasts into induced neural stem cell-like cells (ciNSLCs) using a cocktail of nine components (M9). The resulting ciNSLCs closely resemble primary neural stem cells molecularly and functionally. Transcriptome analysis revealed that M9 induces a gradual and specific conversion of fibroblasts toward a neural fate. During reprogramming specific transcription factors such as Elk1 and Gli2 that are downstream of M9-induced signaling pathways bind and activate endogenous master neural genes to specify neural identity. Our study provides an effective chemical approach for generating neural stem cells from mouse fibroblasts and reveals mechanistic insights into underlying reprogramming processes.