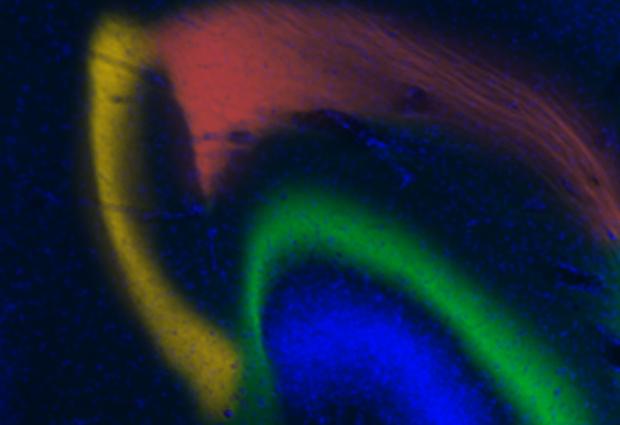
Pictured is an artist’s interpretation of neurons firing in sporadic, coordinated bursts. “By having these different bursts coming at different moments in time, you can keep different items in memory separate from one another,” Earl Miller says. (credit: Jose-Luis Olivares/MIT)
Think of a sentence you just read. Like that one. You’re now using your working memory, a critical brain system that’s roughly analogous to RAM memory in a computer.
Neuroscientists have believed that as information is held in working memory, brain cells associated with that information must be firing continuously. Not so — they fire in sporadic, coordinated bursts, says Earl Miller, the Picower Professor in MIT’s Picower Institute for Learning and Memory and the Department of Brain and Cognitive Sciences.
That makes sense. These different bursts could help the brain hold multiple items in working memory at the same time, according to the researchers. “By having these different bursts coming at different moments in time, you can keep different items in memory separate from one another,” says Miller, the senior author of a study that appears in the March 17 issue of Neuron.
Bursts of activity, not averaged activity
So why hasn’t anyone noticed this before? Because previous studies averaged the brain’s activity over seconds or even minutes of performing the task, Miller says. “We looked more closely at this activity, not by averaging across time, but from looking from moment to moment. That revealed that something way more complex is going on.”
To do that, Miller and his colleagues recorded neuron activity in animals as they were shown a sequence of three colored squares, each in a different location. Then, the squares were shown again, but one of them had changed color. The animals were trained to respond when they noticed the square that had changed color — a task requiring them to hold all three squares in working memory for about two seconds.
The researchers found that as items were held in working memory, ensembles of neurons in the prefrontal cortex were active in brief bursts, and these bursts only occurred in recording sites in which information about the squares was stored. The bursting was most frequent at the beginning of the task, when the information was encoded, and at the end, when the memories were read out.
The findings fit well with a model that Lundqvist had developed as an alternative to the model of sustained activity as the neural basis of working memory. According to the new model, information is stored in rapid changes in the synaptic strength of the neurons. The brief bursts serve to “imprint” information in the synapses of these neurons, and the bursts reoccur periodically to reinforce the information as long as it is needed.
The bursts create waves of coordinated activity at the gamma frequency (45 to 100 hertz), like the ones that were observed in the data. These waves occur sporadically, with gaps between them, and each ensemble of neurons, encoding a specific item, produces a different burst of gamma waves, like a fingerprint.
Implications for other cognitive functions
The findings suggest that it would be worthwhile to look for this kind of cyclical activity in other cognitive functions such as attention, the researchers say. Oscillations like those seen in this study may help the brain to package information and keep it separate so that different pieces of information don’t interfere with each other.
Robert Knight, a professor of psychology and neuroscience at the University of California at Berkeley, says the new study “provides compelling evidence that nonlinear oscillatory dynamics underlie prefrontal dependent working memory capacity.”
“The work calls for a new view of the computational processes supporting goal-directed behavior,” adds Knight, who was not involved in the research. “The control processes supporting nonlinear dynamics are not understood, but this work provides a critical guidepost for future work aimed at understanding how the brain enables fluid cognition.”
editor’s comments: I’m curious how this relates to forgetting things to make space to learn new things. (Turns out the hippocampus works closely with the prefrontal cortex in working memory, as this open-access Nature paper explains.) Also, what’s the latest on how many things we can keep in working memory (it used to be around five)? Is that number limited by forgetting or by the capacity to differentiate different spike trains? Any tricks for keeping more things in working memory?
Abstract of Gamma and Beta Bursts Underlie Working Memory
Working memory is thought to result from sustained neuron spiking. However, computational models suggest complex dynamics with discrete oscillatory bursts. We analyzed local field potential (LFP) and spiking from the prefrontal cortex (PFC) of monkeys performing a working memory task. There were brief bursts of narrow-band gamma oscillations (45–100 Hz), varied in time and frequency, accompanying encoding and re-activation of sensory information. They appeared at a minority of recording sites associated with spiking reflecting the to-be-remembered items. Beta oscillations (20–35 Hz) also occurred in brief, variable bursts but reflected a default state interrupted by encoding and decoding. Only activity of neurons reflecting encoding/decoding correlated with changes in gamma burst rate. Thus, gamma bursts could gate access to, and prevent sensory interference with, working memory. This supports the hypothesis that working memory is manifested by discrete oscillatory dynamics and spiking, not sustained activity.