
Very few science fiction stories have perfect science in them. Although they have some problems, the stories can still be used to explain cool science.
The post The Science in The Martian Isn’t Perfect, But That’s OK appeared first on WIRED.

Science and reality

Very few science fiction stories have perfect science in them. Although they have some problems, the stories can still be used to explain cool science.
The post The Science in The Martian Isn’t Perfect, But That’s OK appeared first on WIRED.

Reconstituting epithelial (skin) microtissues with programmed size, shape, composition, spatial heterogeneity, and embedding extracellular matrix. Scheme and images of fully embedded aggregates of human luminal and myoepithelial cells. (credit: Michael E Todhunter et al./Nature Methods)
A new technique developed by UCSF scientists for building organoids (tiny models of human tissues) more precisely turns human cells into the biological equivalent of LEGO bricks. Called DNA Programmed Assembly of Cells (DPAC), it allows researchers in hours to create arrays of thousands of custom-designed organoids, such as models of human mammary glands containing several hundred cells each.
These mini-tissues in a dish can be used to study how particular structural features of tissue affect normal growth or go awry in cancer. They could be used for therapeutic drug screening and to help teach researchers how to grow whole human organs.
The new technique, reported in an open-the journal Nature Methods on Aug. 31, allows for researchers to “take any cell type we want and program just where it goes,” said Zev Gartner, PhD, the paper’s senior author and an associate professor of pharmaceutical chemistry at UCSF. “We can precisely control who’s talking to whom and who’s touching whom at the earliest stages.”
There are very few limits to the tissues this technology can mimic, he said. “One potential application would be that within the next couple of years, we could be taking samples of different components of a cancer patient’s mammary gland and building a model of their tissue to use as a personalized drug screening platform. Another is to use the rules of tissue growth we learn with these models to one day grow complete organs.”
Studying how the cells of complex tissues like the mammary gland self-organize, make decisions as groups, and break down in disease has been a challenge to researchers. The living organism is often too complex to identify the specific causes of a particular cellular behavior. On the other hand, cells in a dish lack the critical element of realistic 3-D structure.
DNA as molecular Velcro and bar code
To specify the 3-D structure of their organoids, the researchers incubate cells with tiny snippets of single-stranded DNA engineered to slip into the cells’ outer membranes, covering each cell like the hairs on a tennis ball. These DNA strands act both as a sort of molecular Velcro and as a bar code that specifies where each cell belongs within the organoid. When two cells incubated with complementary DNA strands come in contact, they stick fast. If the DNA sequences don’t match, the cells float on by. Cells can be incubated with several sets of DNA bar codes to specify multiple allowable partners.

A whole-mount image of a digitized mouse mammary fat pad (reproduced with permission of W. Muller) used to print a pattern of DNA spots, and rendered as a 1.6-cm-long pattern of single cells fully embedded in gelatinous protein mixture (credit: Michael E Todhunter et al./Nature Methods)
To turn these cellular LEGOs into arrays of organoids that can be used for research, Gartner’s team lays down the cells in layers, with multiple sets of cells designed to stick to particular partners. This lets them build up complex tissue components like the mammary gland. It also lets them experiment with specifically adding in a single cell with a known cancer mutation to different parts of the organoid to observe its effects.
To demonstrate the precision of the technique and its ability to generalize to many different human tissue types, the research team created several proof-of-principle organoid arrays mimicking human tissues such as branching vasculature and mammary glands.
In one experiment, the researchers created arrays of mammary epithelial cells and asked how adding one or more cells expressing low levels of the cancer gene RasG12V affected the cells around them. They found that normal cells grow faster when in an organoid with cells expressing RasG12V at low levels, but required more than one mutant cell to kick-start this abnormal growth. They also found that placing cells with low RasG12V expression at the end of a tube of normal cells allowed the mutant cells to branch and grow, drawing normal cells behind them like a bud at the tip of a growing tree branch.
Gartner’s group plans to use the technique to investigate what cellular or structural changes in mammary glands can lead to the breakdown of tissue architecture associated with tumors that metastasize, invading other parts of the body and threatening the life of the patient. They also hope to use what they learn from simple models of different tissue types to ultimately build functional human tissues like lung and kidney and neural circuits using larger-scale techniques.
Abstract of Programmed synthesis of three-dimensional tissues
Reconstituting tissues from their cellular building blocks facilitates the modeling of morphogenesis, homeostasis and disease in vitro. Here we describe DNA-programmed assembly of cells (DPAC), a method to reconstitute the multicellular organization of organoid-like tissues having programmed size, shape, composition and spatial heterogeneity. DPAC uses dissociated cells that are chemically functionalized with degradable oligonucleotide ‘Velcro’, allowing rapid, specific and reversible cell adhesion to other surfaces coated with complementary DNA sequences. DNA-patterned substrates function as removable and adhesive templates, and layer-by-layer DNA-programmed assembly builds arrays of tissues into the third dimension above the template. DNase releases completed arrays of organoid-like microtissues from the template concomitant with full embedding in a variety of extracellular matrix (ECM) gels. DPAC positions subpopulations of cells with single-cell spatial resolution and generates cultures several centimeters long. We used DPAC to explore the impact of ECM composition, heterotypic cell-cell interactions and patterns of signaling heterogeneity on collective cell behaviors.
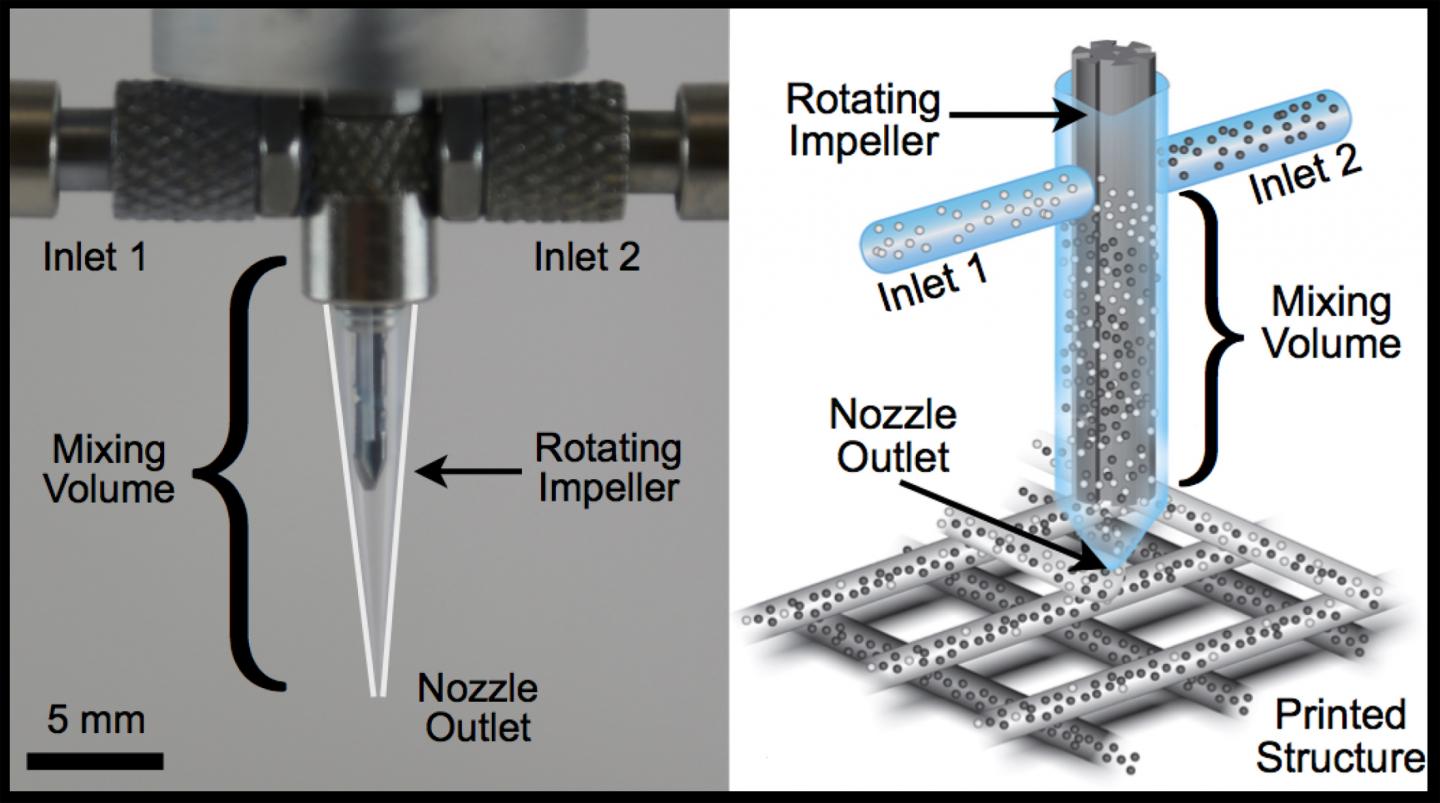
Multiple-materials printer. Each fluid enters the mixing chamber through a separate inlet and is mixed in a narrow gap by an impeller rotating at a constant rate. Optical image (left) and schematic illustration (right) of impeller-based mixing nozzle. (credit: Thomas Ober, Harvard SEAS/Wyss Institute)
Harvard researchers have designed new printheads for 3-D printers that can simultaneously handle multiple materials with different properties, allowing for 3-D printing wearable devices, flexible electronics, and soft robots.
To print a flexible device, including the electronics, a 3-D printer must be able to seamlessly transition from a flexible material that moves with the wearer’s joints for wearable applications, to a rigid material that accommodates the electronic components. It would also need to be able to embed electrical circuitry using multiple inks of varying conductivity and resistivity, and precisely switching between them while changing composition and geometry. And do it all in real time.
How this will change 3-D printing
The researchers say they have designed a new multimaterial printhead that do all of the above. It can handle a wide range of complex fluids by using a rotating impeller inside a microscale nozzle, seamlessly printing combinations of materials and processes that were not formerly possible:
The research was led by Jennifer A. Lewis, the Hansjörg Wyss Professor of Biologically Inspired Engineering at the Harvard John A. Paulson School of Engineering and Applied Sciences (SEAS) and a Core Faculty Member at the Wyss Institute for Biologically Inspired Engineering at Harvard. The work was published in an open-access paper in Proceedings of the National Academy of Sciences (PNAS). It was supported by the Department of Energy Energy Frontier Research Center on Light-Material Interactions in Energy Conversion, the Intelligence Community Postdoctoral Fellowship program, and the Society in Science Branco-Weiss Foundation.
“The recent work by the Lewis Group is a significant advancement to the field of additive manufacturing,” said Christopher Spadaccini, Director of the Center for Engineered Materials, Manufacturing and Optimization at Lawrence Livermore National Lab. “By allowing for the mixing of two highly viscous materials on the fly, the promise of mixed material systems with disparate mechanical and functional properties becomes much more realistic. Before, this was really only a concept. This work will be foundational for applications which [require] integrated electrical and structural materials.”
Abstract of Active mixing of complex fluids at the microscale
Mixing of complex fluids at low Reynolds number is fundamental for a broad range of applications, including materials assembly, microfluidics, and biomedical devices. Of these materials, yield stress fluids (and gels) pose the most significant challenges, especially when they must be mixed in low volumes over short timescales. New scaling relationships between mixer dimensions and operating conditions are derived and experimentally verified to create a framework for designing active microfluidic mixers that can efficiently homogenize a wide range of complex fluids. Active mixing printheads are then designed and implemented for multimaterial 3D printing of viscoelastic inks with programmable control of local composition.

Reports of problems in women who use Essure, a form of permanent birth control, are skyrocketing thanks to a group on Facebook.
The post How a Facebook Group Got the FDA to Reconsider a Type of Birth Control appeared first on WIRED.