
No matter how wet the winter, California farms pump more water than rainfall can replace.
The post Devastating Chart Shows Why El Niño Won’t Fix the Drought appeared first on WIRED.

Science and reality

No matter how wet the winter, California farms pump more water than rainfall can replace.
The post Devastating Chart Shows Why El Niño Won’t Fix the Drought appeared first on WIRED.

This video shows a very massive pressurized vessel being transported by a giant trailer. Why is the trailer so large? Why are there so many trucks?
The post How on Earth Could Trucks Move This 1,000-Ton Load? appeared first on WIRED.

Liquid ports carry cooling water to specially designed passages etched into the backs of FPGA devices to provide more effective cooling. The liquid cooling provides a significant advantage in computing throughput. (credit: Rob Felt/Georgia Tech)
Using microfluidic passages cut directly into the backsides of production field-programmable gate array (FPGA) devices, Georgia Institute of Technology researchers have put liquid cooling where it’s needed the most: a few hundred microns away from where the transistors are operating.
Combined with connection technology that operates through structures in the cooling passages, the new technologies could allow development of denser and more powerful integrated electronic systems that would no longer require heat sinks or cooling fans on top of the integrated circuits.
Working with popular 28-nanometer FPGA devices made by Altera Corp., the researchers demonstrated a monolithically-cooled chip that can operate at temperatures more than 60 percent below those of similar air-cooled chips.
The lower temperatures can also mean longer device life and less current leakage. The cooling comes from simple de-ionized water flowing through microfluidic passages that replace the massive air-cooled heat sinks normally placed on the backs of chips.
Supported by the Defense Advanced Research Projects Agency (DARPA), the research is believed to be the first example of liquid cooling directly on an operating high-performance CMOS chip.
Liquid cooling has been used to address the heat challenges facing computing systems whose power needs have been increasing. However, existing liquid cooling technology removes heat using cold plates externally attached to fully packaged silicon chips — adding thermal resistance and reducing the heat-rejection efficiency.
In multiple tests, a liquid-cooled FPGA was operated using a custom processor architecture provided by Altera. With a water inlet temperature of approximately 20 degrees Celsius and an inlet flow rate of 147 milliliters per minute, the liquid-cooled FPGA operated at a temperature of less than 24 degrees Celsius, compared to an air-cooled device that operated at 60 degrees Celsius.
The research team chose FPGAs for their test because they provide a platform to test different circuit designs, and because FPGAs are common in many market segments, including defense. However, the same technology could also be used to cool CPUs, GPUs and other devices such as power amplifiers, according to the researchers.
In addition to improving overall cooling, the system could reduce hotspots in circuits by applying cooling much closer to the power source. Eliminating the heat sink could also allow more compact packaging of electronic devices.
The cooling research was funded by DARPA’s Microsystems Technology Office, through the ICECOOL program.
Abstract of Embedded Cooling Technologies For Densely Integrated Electronic Systems
In modern integrated systems, interconnect and thermal management technologies have become two major limitations to system performance. In this paper we present a number of technologies to address these challenges. First, low-loss polymer-embedded vias are demonstrated in thick wafers compatible with microfluidics. Next, fluidic I/Os for delivery of fluid to microfluidic heat sinks are demonstrated in assembled 2.5D and 3D stacks. Next, thermal coupling between dice in 2.5D and 3D systems is explored. Lastly, the utility of microfluidic cooling is demonstrated through an FPGA, built in a 28nm process, with a monolithically integrated microfluidic heat sink.

Astroglial cells before (top) and after (bottom) treatment with small-molecule cocktails (credit: Gong Chen lab, Penn State University)
Researchers have succeeded in transforming human support brain cells, called astroglial cells, into functioning neurons for brain repair.
The new technology opens the door to future development of drugs that patients could take as pills to regenerate neurons and to restore brain functions lost after traumatic injuries, stroke, or diseases such as Alzheimer’s.
Previous research, such as conventional stem-cell therapy, has required brain surgery, so it is much more invasive and prone to immune-system rejection and other problems.
The new research, led by Gong Chen, Professor of Biology and the Verne M. Willaman Chair in Life Sciences at Penn State University, was published online today (Oct. 15) in the journal Cell Stem Cell.
“We have discovered a cocktail of small molecules that can reprogram human brain astroglial cells into neuron-like cells after eight to ten days of chemical treatment,” Chen said. The reprogrammed nerves survived for more than five months in cell culture, where they formed functional synaptic networks.
The scientists also injected the reprogrammed human neurons into the brains of living mice, where they integrated into the neural circuits and survived there for at least one month.
“The small molecules are not only easy to synthesize and package into drug pills, but also much more convenient for use by patients than other methods now being developed,” Chen said.
Converting astroglial cells into neurons
Astroglial cells surround neurons and provide them with support, protection, oxygen, and nutrients. But when brain tissues are damaged by strokes or trauma, the astroglial cells react by multiplying — sometimes so much that they clog up the nervous system by forming a scar. These astroglial scars — a difficult research challenge for many decades — can cause health problems by preventing nerve regeneration and by blocking nerve-to-nerve communications between different regions of the brain.
Chen’s group previously invented a method to convert astroglial cells into neurons using viral particles. But Chen also wanted to investigate whether small chemical compounds, which could be packaged into swallowable pills, could also do the job.
Five students on Chen’s research team, led by graduate student Lei Zhang, tested hundreds of different conditions and eventually identified a cocktail of small molecules that can convert human astroglial cells into functional neurons in a cell-culture dish in the laboratory. The students found that adding small molecules in a certain sequence transformed the cultured human astroglial cells from a flat, polygon shape into a neuron-like shape with long “arms” called axons and dendrites.
“These chemically generated neurons are comparable to normal brain neurons in terms of firing electric activity and release of neurotransmitters,” Chen said. “Importantly, the human astroglial-converted neurons survived longer than five months in cell culture and longer than one month in the living mouse brain after transplantation.”
Chen acknowledges that further development, laboratory testing, and a series of clinical trials are still required, but he hopes that this new technology may have broad applications in the future treatment of stroke, Alzheimer’s disease, Parkinson’s disease, and other neurological disorders.
“Our dream is that, one day, patients with brain disorders can take drug pills at home to regenerate neurons inside their brains without any brain surgery and without any cell transplantation,” he said.
Scientists from Emory University School of Medicine were also involved in the research.
Abstract of Small Molecules Efficiently Reprogram Human Astroglial Cells into Functional Neurons
We have recently demonstrated that reactive glial cells can be directly reprogrammed into functional neurons by a single neural transcription factor, NeuroD1. Here we report that a combination of small molecules can also reprogram human astrocytes in culture into fully functional neurons. We demonstrate that sequential exposure of human astrocytes to a cocktail of nine small molecules that inhibit glial but activate neuronal signaling pathways can successfully reprogram astrocytes into neurons in 8-10 days. This chemical reprogramming is mediated through epigenetic regulation and involves transcriptional activation of NEUROD1 and NEUROGENIN2. The human astrocyte-converted neurons can survive for >5 months in culture and form functional synaptic networks with synchronous burst activities. The chemically reprogrammed human neurons can also survive for >1 month in the mouse brain in vivo and integrate into local circuits. Our study opens a new avenue using chemical compounds to reprogram reactive glial cells into functional neurons.
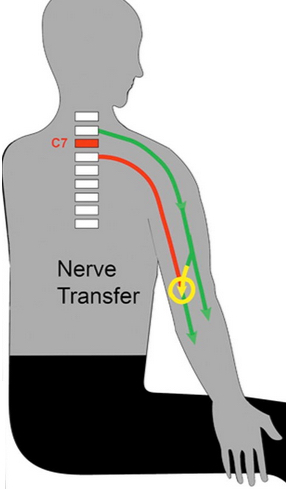
A nerve transfer bypasses the zone of a spinal cord injury (C7). Functional nerves (green) that are under volitional control are rerouted (yellow) to nerves (red) that come off below the spinal cord injury. (credit: Washington University in St. Louis)
A pioneering surgical technique has restored some hand and arm movement to nine patients immobilized by spinal cord injuries in the neck, reports a new study at Washington University School of Medicine in St. Louis.
Bypassing the spinal cord, the surgeons rerouted healthy nerves sitting above the injury site, usually in the shoulders or elbows, to paralyzed nerves in the hand or arm. Once a connection was established, patients underwent extensive physical therapy to train the brain to recognize the new nerve signals, a process that takes about 6–18 months.
The technique targets patients with injuries at the C6 or C7 vertebra, the lowest bones in the neck. It typically does not help patients who have lost all arm function due to higher injuries in vertebrae C1 through C5.
“Physically, nerve-transfer surgery provides incremental improvements in hand and arm function. However, psychologically, these small steps are huge for a patient’s quality of life,” said the study’s lead author, Ida K. Fox, MD, assistant professor of plastic and reconstructive surgery.
One of the most humbling effects of spine damage is the inability to manage bladder or bowel functions. “People with spinal cord injuries cannot control those functions because their brains can’t talk to the nerves in the lower body,” said Fox, who performs surgeries at Barnes-Jewish Hospital.
The study is published in an open-access paper in the October issue of the American Society of Plastic Surgeons’ journal, Plastic and Reconstructive Surgery.
Ultimately, medical professionals hope to discover a way to restore full movement to the estimated 250,000 people in the U.S. living with spinal cord injuries. More than half of such injuries involve the neck. However, until a cure is found, progress in regaining basic independence in routine tasks is important.
Abstract of Nerve transfers to restore upper extremity function in cervical spinal cord injury: Update and preliminary outcomes
Background: Cervical spinal cord injury can result in profound loss of upper extremity function. Recent interest in the use of nerve transfers to restore volitional control is an exciting development in the care of these complex patients. In this article, the authors review preliminary results of nerve transfers in spinal cord injury.
Methods: Review of the literature and the authors’ cases series of 13 operations in nine spinal cord injury nerve transfer recipients was performed. Representative cases were reviewed to explore critical concepts and preliminary outcomes.
Results: The nerve transfers used expendable donors (e.g., teres minor, deltoid, supinator, and brachialis) innervated above the level of the spinal cord injury to restore volitional control of missing function such as elbow extension, wrist extension, and/or hand function (posterior interosseous nerve or anterior interosseous nerve/finger flexors reinnervated). Results from the literature and the authors’ patients (after a mean postsurgical follow-up of 12 months) indicate gains in function as assessed by both manual muscle testing and patients’ self-reported outcomes measures.
Conclusions: Nerve transfers can provide an alternative and consistent means of reestablishing volitional control of upper extremity function in people with cervical level spinal cord injury. Early outcomes provide evidence of substantial improvements in self-reported function despite relatively subtle objective gains in isolated muscle strength. Further work to investigate the optimal timing and combination of nerve transfer operations, the combination of these with traditional treatments (tendon transfer and functional electrical stimulation), and measurement of outcomes is imperative for determining the precise role of these operations.

The orbiter captured illuminated images of the moon's jagged fractures and deep craters.
The post Check Out These Awesome Images of Enceladus from Cassini appeared first on WIRED.

We ran the numbers. While American women’s average BMI has gone up, Playboy’s largely imagined BMI has gone down.
The post Playboy’s Image of the Ideal Woman Sure Has Changed appeared first on WIRED.

It's been three months since New Horizons flew through the Pluto system, and the science it sent back finally went through peer review.
The post Pluto’s Flyby Data Finally Enters the Scientific Record appeared first on WIRED.